RAINWATER METALS
Clifford E Carnicom
Jul 27 2001
Extraordinary levels of metallic particulates are now in the process of being identified within rainwater samples. The particular sample shown here is from rainwater collected on Jul 26 2001 in Santa Fe NM. Distillation of rainwater is being used as the method to accumulate the metallic particulates which are now readily visible to the naked eye within a test tube under sufficient lighting. Methods of crystalline chemistry have previously been outlined; in the case shown here no chemical reagents are involved. The high levels of metallic particulates are directly visible in concentrated form after distillation occurs.
The method shown on this page uses an initial sample of approximately 30ml of rainwater in an Erlenmeyer flask. This amount of rainwater is distilled to leave a concentrate remainder of approximately 4ml. This sample is illuminated with a strong source of light and subsequently photographed.
Future analysis will submit these samples to microscopic examination.
A video file (.mpg format, ~2meg. in size, 36secs. duration) of the current analysis is available for download at the end of this page. A file of this size and resolution is required to adequately demonstrate the reflective and insoluble nature of the material.
Notes : August 1 2001:
The pH of the concentrated rainwater samples has recently been measured at 7.6 or higher (end of scale currently available). This demonstrates a level of alkalinity much higher than that expected in rainwater. The equlibrium pH of rainwater is approximately 5.6 due to the presence of carbonic acid; it is usually less in industrialized areas due to the well-known acid rain phenomenon.
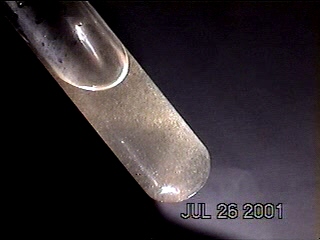
Distilled Concentrate of Rain Water in Test Tube.
Metallic particulates are highly visible.

Distilled Concentrate of Rain Water in Test Tube.
Metallic particulates are highly visible.
Video File of Current Analysis
(.mpg format, 36secs., 2meg)
Click Here to Download
(Windows : To Save File to Hard Drive :
Right Click, Save Link As)
(Windows Media Player Option :
Use ALT+ENTER to make full screen size)
[File missing as of 2016/02/09]



