Morgellons:
Unique Protein Isolated & Characterized
by
Clifford E Carnicom
Aug 13 2017
Edited Oct 01 2017
Note: Carnicom Institute is not offering any medical advice or diagnosis with the presentation of this information. CI is acting solely as an independent research entity that is providing the results of extended observation and analysis of unusual biological conditions that are evident. Each individual must work with their own health professional to establish any appropriate course of action and any health related comments in this paper are solely for informational purposes.
A protein generated by the microorganism associated with the Morgellons condition (tentatively classified in past research as a “cross-domain bacteria”, i.e., CDB) has been isolated and characterized in several ways. There is little doubt that this protein is at the heart of the physiological and biochemical changes that occur within the body by those affected. Related research has been conducted with success for some time, however, the recent work represents a different and separate approach from previous accomplishments. Proteins are at the crux of biochemistry and biological research, and they have great importance in relation to biological structure. There are usually numerous applications (beyond health aspects alone) that develop with the advent of a new or isolated protein, and it is expected that the current work can eventually follow this suit.
Only the general nature of the protein will be described at this point. The protein is organometallic in nature, highly water soluble, and strongly acidic. Additional resources of significance and support from the health communities will be required to develop the series of discoveries into tangible benefits.
Some of the methods that been employed to define the unique nature and characteristics of the protein include:
- The molecular weight of the protein has been estimated with laboratory methods.
- The solubility and polarity of the protein has been assessed.
- Pyrolysis with gas chromatography (GC) has been applied to the protein to examine its thermal decomposition into various subcomponents.
- Headspace methods have been used to examine the nature and volatility of gaseous metabolism of the microorganism.
- Infrared (IR) analysis has been used to identify the primary functional groups of the protein, along with the analysis of various GC trapped components.
- Ultraviolet (UV) analysis of the protein has been conducted.
- Candidate amino acid composition, at least to a partial extent, has been established.
- The pH of the protein has been measured.
- The isolectric point of the protein has been determined via titration.
- Precipitation methods for the protein have been developed.
- A metallic nature of the protein has been verified.
- The index of refraction for the protein has been determined by measurement.
- A concentration-dilution model for the protein has been developed based upon the index of refraction.
- The polarimetric nature of the protein has been examined.
- The electrical conductivity of the protein as a function of concentration and dilution has been determined.
- The Oxidation Reduction Potential (OPR) of the dilute protein has been measured.
- A colorimetric test for the existence of the protein has been established.
- Initial molecular models proposals have been established for some of the simpler components of the headspace-pyrolysis components with GC – IR coupling.
- Initial anticipated impacts upon physiology, i.e., absorption levels, are under investigation.
- The Bradford reagent identification test for protein identification has been applied via visible light spectroscopy.
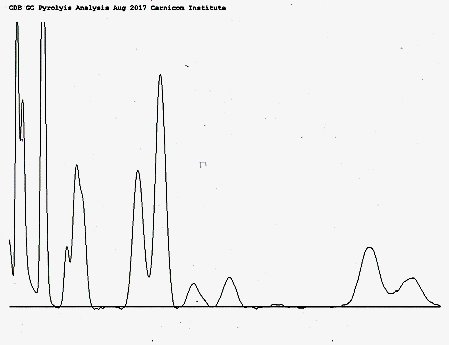
GC Pyrolysis Chromatogram of Numerous Components of CDB Isolated Protein
(significant hydrocarbon structure is identified within)
The isolation and characterization of this particular protein and its properties are of importance and uniqueness in the research related to the Morgellons condition. The attributes identified are numerous and specific to the microorganism that has been extensively identified, examined and researched. The uniqueness of the protein is essentially guaranteed. The method of development of the protein also represents a distinct and recent advance in the history of CI research, and it is hoped at some point that the work will be placed to the advantage and benefit of the public.
Clifford E Carnicom
Aug 11 2017
Edited Oct 01 2017
Born Clifford Bruce Stewart
Jan 19 1953



