The Discovery of Thiocyanates within the Cross-Domain Bacteria
by
Clifford E Carnicom
Jun 25 2018
Edit Aug 06 2018
The chemical signature of a significant and potentially toxic compound has been repeatedly identified and confirmed within the cross-domain bacteria (CDB – CI, May 2014) that are causative within the Morgellons condition. This chemical structure does and will exist as another unique biochemical marker of this particular microorganism.
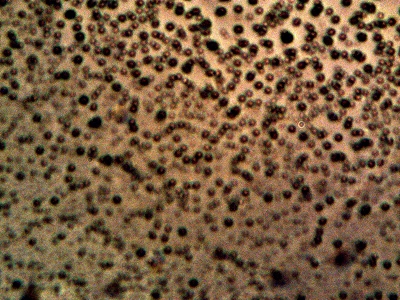
Microphotograph of the Cross-Domain Bacteria
Isolated May 2014
Magnification Approx. 5000x
The chemical functional group identified is that of an isothiocyanate. This chemical, when in excess within the body, does pose significant health and toxicity risks. Areas of special concern include those of thyroid inhibition, liver toxicity and tinnitus; there are numerous other health issues that arise or relate to these as well.
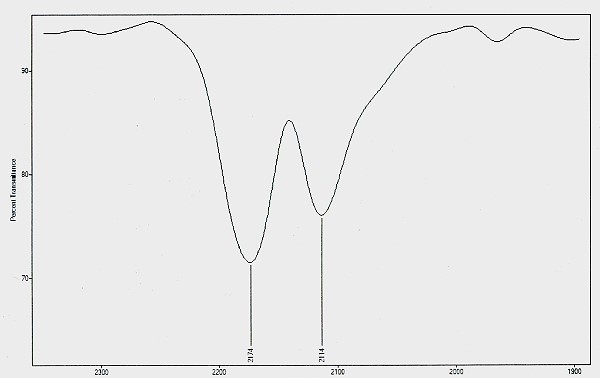
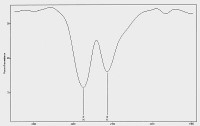
Infrared spectrum of the isothiocyanate chemical structure existing within the CDB
The method of detection involves the use of pyrolysis and infrared spectrometry. The relevant portion of the spectrum is shown in the plot above, with absorption peaks of 2174 cm-1 and 2114 cm-1. The double absorption peak in this region offers further uniqueness in the identification process.
The general description of the thiocyanate and isothiocyanate functional groups can be located within most organic chemistry reference sources. A typical partial entry is of the following form:
Thiocyanate is the anion [SCN]−. Common derivatives include the colourless salts potassium thiocyanate and sodium thiocyanate. Organic compounds containing the functional group SCN are also called thiocyanates. Mercury(II) thiocyanate was formerly used in pyrotechnics.
Thiocyanate is produced by the reaction of elemental sulfur or thiosulfate with cyanide.
Organic and transition metal derivatives of the thiocyanate ion can exist as “linkage isomers”. In thiocyanates, the organic group (or metal ion) is attached to sulfur: R−S−C≡N has a S–C single bond and a C≡N triple bond. In isothiocyanates, the substituent is attached to nitrogen: R−N=C=S has a S=C double bond and a C=N double bond.
(https://en.wikipedia.org/wiki/Thiocyanate)
The more lay interpretation of one of the more immediate concerns is as follows:
“The overall effect of thiocyanate is to hamper utilization of iodide, the main effect of thiocyanate is to worsen iodine deficiency. By this mechanism thiocyanate is one of the most important environmental compounds influencing the occurrence of thyroid disease.”
(https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/thiocyanate)
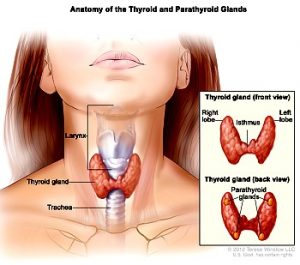
The Thyroid Gland
“The thyroid gland is a small, butterfly-shaped gland located in the base of the neck just below the Adam’s apple.
Although relatively small, the thyroid gland plays a huge role in our body, influencing the function of
many of the body’s most important organs, including the heart, brain, liver, kidneys and skin.”
(thyroidawareness.com)
(image source: Oregon Health & Science University)
It is appropriate to mention that the existence of thiocyanates and isothiocyanates, in and of itself, does not necessarily indicate that a toxicity situation exists. It appears to be a matter of degree and concentration within the body. There are many natural sources of thiocyanates, especially the sulfur-containing foods such as cabbage, turnips, broccoli, Brussels sprouts, kale and cauliflower (Brassica vegetables). One may even find thiocyanates sold in supplement form for the diet. There are also some studies that indicate anti-cancerous properties of thiocyanates.
On the other hand, there is a preponderance of evidence in the literature that an excess of thiocyanates most certainly presents significant health risks and serious toxicity issues.
For example, a toxicity review of the issue will quickly reveal statements such as the following:
1. From the International Cyanide Management Code:
“Although thiocyanate is approximately seven times less toxic than cyanide, increased thiocyanate concentrations …can adversely affect the thyroid.”
(https://www.cyanidecode.org/cyanide-facts/environmental-health-effects)
2. From the National Institutes of Health:
“Thiocyanate [SCN-] is a complex anion which is a potent inhibitor of iodide transport. It is the detoxification product of cyanide and can easily be measured in body fluids. Consumption of naturally occurring goitrogens, certain environmental toxins and cigarette smoke can significantly increase SCN- concentrations to levels potentially capable of affecting the thyroid gland.”
(https://www.ncbi.nlm.nih.gov/pubmed/14757960)
3. From Biochemistry & Pharmacology: Open Access:
“Many agents in the environment interfere with thyroid gland morphology and function acting directly on the gland or indirectly by altering the regulatory mechanism of thyroid gland. The uptake and utilization of iodine, by the thyroid gland is impaired by the pseudo halide thiocyanate (SCN-). Thiocyanate is formed from cyanogenic substances. It is metabolized in thyroid gland. The role of thiocyanate ion in the homeostasis of thyroid is a provocative issue where IDD [Iodine Deficiency Disorder] persists in spite of adequate iodine intake and consumption of cyanogenic plant food is relatively high.”
(https://www.omicsonline.org/open-access/iodine-thiocyanate-and-the-thyroid-2167-0501-1000171.php?aid=54337)
4. From the T3DB Toxin Database:
“Thiocyanates are known to affect the thyroid glands, reducing the ability of the gland to produce hormones that are necessary for the normal function of the body.”
http://www.t3db.ca/toxins/T3D0089#
and the list continues. Additional relationships with liver function are also simple to establish:
1. From McKendree University:
“The ingestion of this [tobacco] smoke leads to the formation of the thiocyanate ion (SCN-) in the liver.”
https://www.mckendree.edu/academics/scholars/issue1/krebs.htm
2. From the Anatomy Journal of Africa:
“The use of thiocyanate as an anti-sickling drug is currently on the increase among sickle cell disease (SCD) patients. The continuous use of this substance without sufficient toxicity data does not guaranty continuously functional and healthy internal organs among the SCD patients that are susceptible to multi-organ failure such as hepatic failure.
…Therefore, this study can be used to infer that SCN use in sickle cell disease management regimen can cause hepatocellular damage in wistar rats.”
(https://www.ajol.info/index.php/aja/article/view/135925)
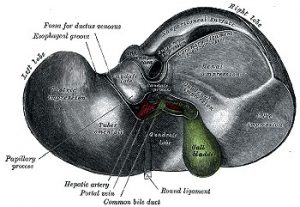
The Liver
“The liver plays a central role in all metabolic processes in the body.”
(https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0072577/)
(image source: commons.wikimedia.org)
This investigative line of research can continue as well.
If we accept that there are indeed toxicity issues that result from an excess of thiocyanates-isothiocyanates in the body, our attention naturally will turn to whether methods exist to reduce this compound.
There will, of course, be an immediate interest in what can be done to assist the thyroid. Before discussing that topic further, however, it may be of benefit to pay attention to this single line of text from a paper at the National Cancer Institute:
“In addition, several case-control studies have shown that specific forms of the gene that encodes glutathione S-transferase, which is the enzyme that metabolizes and helps eliminate isothiocyanates from the body, may influence the association between cruciferous vegetable intake and human lung and colorectal cancer risk”
(https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/cruciferous-vegetables-fact-sheet)
This is certainly a most interesting statement, as it returns us to the prospective benefits of gluathione precursors, anti-oxidants, vitamins, and the tremendous importance of glutathione itself. These topics have been extensively discussed on this site over the years, and there is no need to repeat that work here. Suffice it to say that attention should be paid to those previous discussions; please recall the difficulties of direct glutathione absorption. Based on the above references to liver health, it is also important to understand that glutathione is an essential metabolic molecule that is produced in the liver of humans and animals.
In addition, the role of selenium acting as a cofactor to assist enzymatic activity in the production of glutathione is also of benefit to understand and realize. Put simply,
“The mineral selenium accelerates the antioxidizing action of glutathione.”
(https://saveourbones.com/glutathione-cofactors-how-they-protect-your-bones-and-how-to-get-them/)
The previous reference also helps as a summary to the numerous cofactors that increase glutathione production. Notice again the numerous references to the suite of vitamins (e.g., B, C, E) within the above article, as well as alpha lipoic acid, each of which has been mentioned extensively on this site with respect to antioxidant benefits.
Furthermore, we note the relationship between selenium and liver health as well:
“Selenium supports the immune system and thyroid function and acts as an antioxidant. It is needed for proper enzymatic activities that protect the body against cancer. It promotes the production of killer T-cells that engulf harmful foreign substances that enter the body and act, in conjunction with vitamin E, as an antioxidant that scavenges the body for free radicals that damage healthy tissue.”
(https://www.livestrong.com/article/441819-selenium-glutathione/)
In closing, for the time being, reference will also be given to a rather detailed and beneficial paper on thyroid health, by Dr. Joseph Bebe, a Board Certified Nutritionist. I think that we may all find it to be a worthwhile read, and I trust that gratitude will be expressed to him for his efforts. I would also request that attention be paid to the extensive list of “Thyroid Suppressing Environmental Chemicals” within, where you will find thiocyanates listed.
The paper is entitled, Reviving Your Thyroid, by Dr. Joseph Bebe.
(http://www.drdebe.com/articles/reviving-your-thyroid)
The subject of this paper has actually been under development over a period of several years. An entire further discussion, at a later time, can take place on the extent of thiocyanate distribution beyond the CDB source. Numerous environmental and biological sample types continue to be examined, and it is fair to say that the thiocyanate issue does not end with the CDB. The joining of metallic ions (such as iron, for example) with isothiocyanate compounds is also a deserving topic of the future. Those additional discussions will have to unfold over time.
The definitive findings of this current report actually bring us full circle to many discussions that have been brought forth on this site over the years. I hope that you will explore those connections with the attention that they deserve, and that your health and the health of those you know and love will benefit from the pursuit.
I must close for now, and I thank you for your time today.
With best regards,
from Clifford
P.S. Here is a side story that may be of interest to readers (If the link remains active):
Rise of Synthetic Biology Means U.S. Government Unprepared for Biowarfare