Clifford E Carnicom
Jan 07 2013
It can now be established with a high degree of certainty that the external casing of the environmental filament samples are composed of keratin or a keratin-like material. This supposition has been in place for a number of years by this researcher; it can now be demonstrated to be the case by direct chemical and spectroscopic means. Certain ramifications of this finding, in conjunction with earlier work, are as follows:
1. It is deduced that the environmental filament is not a naturally occurring material.
2. The filaments contains non-keratin based chemical and biological components within the internals of the filaments. Considerable information regarding the nature of the environmental filaments is available on this site; this information has been accrued over a period of several years of progressive research.
3. The emphasis upon study of the filaments is to be directed to the sub-micron components (biological and chemical) that are internal to the filaments. The keratin aspect of structure is to be interpreted as an encasing mechanism only.
4. The filaments are not hair or spider webs.
5. A false laboratory report has been issued in the past regarding the identification of this filament material (to be discussed in a separate report).
The primary method by which this conclusion has been reached is with chemical and spectroscopic comparison of a known source of keratin with the environmental filament by similiar means. This comparison has been made possible with the recent advance in methods of chemical decomposition of keratin based substances by this researcher. Please see the report entitled “Environmental Filament Penetrated” for this discussion and presentation.
 |
 |
|
| Human Hair | Environmental Filament Sample |
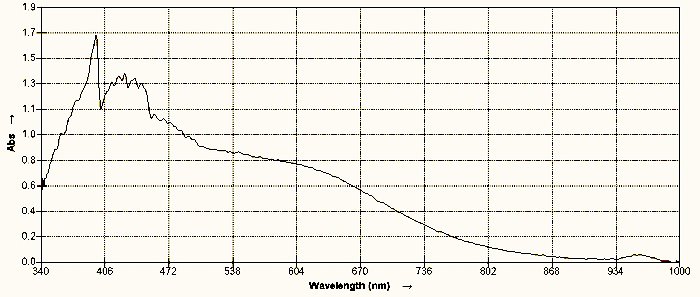
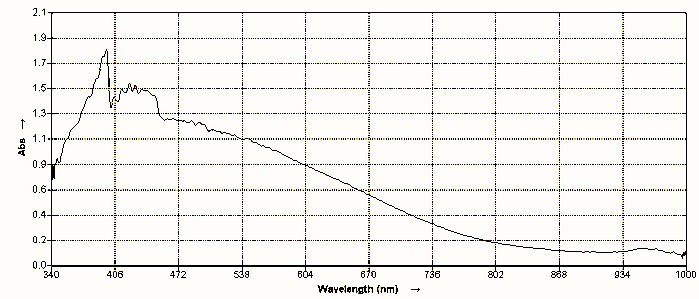
Spectroscopic comparisons of keratin obtained from human hair and the same substance obtained from the environmental filament casing are shown immediately below; it will be seen that they are essentially identical. Additional notes and discussion will follow below the spectra.
| A visual light spectroscopic analysis of keratin obtained from the decomposition of human hair in combination with ninhydrin and heating. Human hair is composed predominantly of keratin. |
| A visual light spectroscopic analysis of the environmental filament after chemical decomposition and in combination with ninhydrin and heat. It will be seen that the spectrum obtained is essentially identical to the keratin spectrum above. The keratin of the environmental filament is interpreted as an encasement structure and it does not account for the biological components that have been repeatedly identified within this protein housing. |
| A photograph of the colored solutions subjected to visual light spectroscopic analysis. The solutions result from chemical decomposition of keratin based structures, in combination with heat and ninyhdrin. The solution on the left is derived from the environmental filament sample; the solution on the right is derived from human hair. Both hues and spectra can vary to some degree by concentration levels within a solution; these examples and spectra indicate a coincidence of relative concentration in each case. |
Additional Notes:
Keratin is an especially impervious protein structure. Observation and study of your own hair is a very good analogy for understanding the hardiness of this particular protein. During the recent trials of study in decomposition, chemical penetration of hair itself represents an excellent example of the challenge of examination of the environmental filaments and their internals. Numerous trials were conducted using strong solutions of sulfuric acid, sodium hydroxide, nitric acid, salicylic acid, sodium hypochlorite (bleach), ammonium thioglycolate and others. All essentially met with failure to the degree needed with the time available. Although some mild success was achieved with a hair sample, the environmental filament sample remained essentially impervious to almost all methods. The best success of decomposition has eventually come forth with the use of a commercial hair declogger used in plumbing systems. This solution is primarily a combination of concentrated sodium hydroxide and concentrated potassium hydroxide. This solutions is highly caustic. The greater success of this method also becomes dependent upon the use of applied heat over an extended time period. It was with the use of this method that valid comparisons, both chemically and spectroscopically, could be made. Considerable work remains before us to acquire the detailed biochemical knowledge of the internal nature of the environmental filaments; this work will continue as the proper resources and equipment avail themselves.