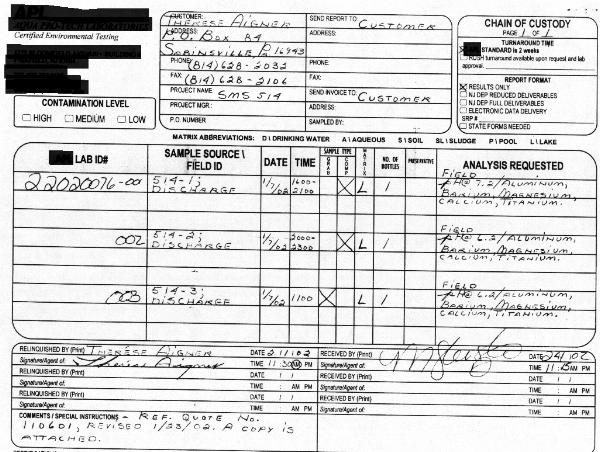
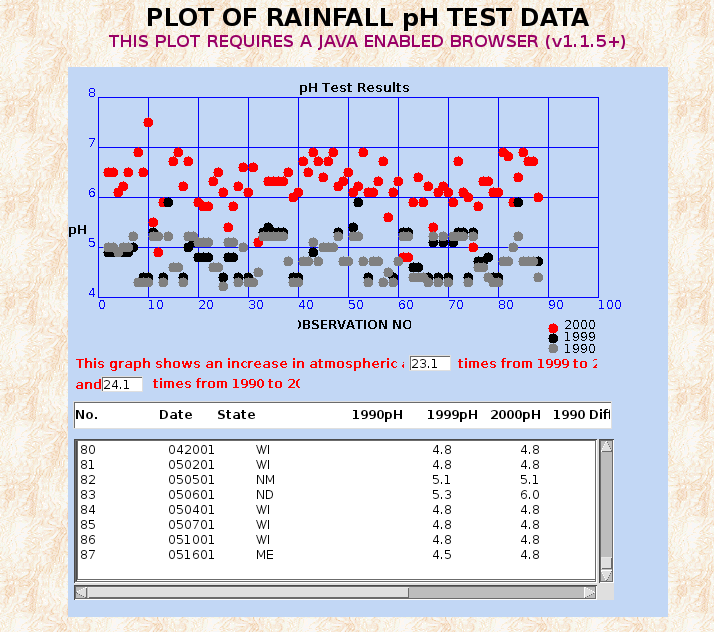
THE EXPECTED COMPOSITION
The following information on the expected composition of the atmosphere is from the book entitled "Introduction to Atmospheric Chemistry", by Peter V. Hobbs, Cambridge University Press, 2000. Please note that the symbols for the chemical elements of Titanium, Aluminum, Barium, Magnesium and Calcium are Ti, Al, Ba, Mg and Ca respectively, and that they are not to be found within this listing. Please also note that concentrations are provided at the parts per trillion level.